Abstract
Background: Sclerotic cGVHD is a highly morbid form of cGVHD, often refractory to standard and secondary therapies and associated with significant decline in patients' quality of life and functioning. We conducted a single-arm phase II multicenter trial to test the hypothesis that ruxolitinib is effective treatment for steroid refractory sclerotic cGVHD (ClinicalTrials.gov ID: NCT03616184).
Methods: Eligibility criteria included adults with cGVHD and skin, joint and/or fascia sclerosis, who had received systemic corticosteroids for >12 months and at least one additional line of therapy, or systemic corticosteroids and at least two additional lines of therapy. Subjects received oral ruxolitinib 10 mg twice daily for 6 months, which was subsequently tapered over another month. Treatment could be extended up to 12 months in patients deriving clinical benefit. The primary objective was to determine the proportion of patients with an NIH-defined response in skin or joints at 6 months. A sample size of 47 patients (estimated 10% drop out) had α of 0.044 and a power of 84% to test the null hypothesis response rate of 25% (Clin Cancer Res. 2016; 22: 319-327) compared to an alternative of 45%. Other endpoints included overall response, non-relapse mortality (NRM), treatment failure (administration of additional systemic therapy, malignancy relapse or mortality), glucocorticoid dose reduction, and patient-reported outcomes (PROs) (Lee symptom scale and Scleroderma Health Assessment Questionnaire).
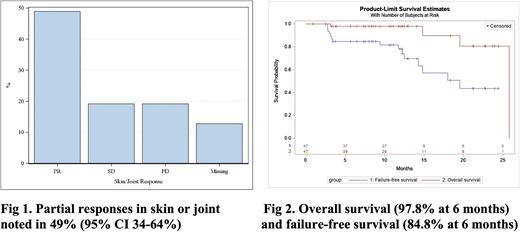
Results: 49 patients were consented between November 2018- June 2021; 2 patients were screen failures. Baseline characteristics of 47 patients included a mean age of 58 (range 23-76) years, 40% female, and 91% white. 87% had peripheral blood as a graft source. 66% had matched unrelated donor transplant. 66% had prior acute GVHD. Median time from cGVHD diagnosis to enrollment was about 48 months. All patients had classic cGVHD at enrollment. Severity included mild (2%), moderate (13%) or severe (85%). Patients had received a mean of 3 prior lines of treatment. Patients received a mean of 8.6 cycles of ruxolitinib. 13% had missing data that precluded response assessment; these patients were considered non-responders. Partial responses (PR) in skin or joint (primary endpoint) were seen in 49% (95% CI 34-64%) at 6 months; other responses included stable disease (19%), progression (19%) and missing (13%) (Fig 1). No complete responses were observed in skin or joint. Overall cGVHD partial responses were seen in 47% (95% CI 32-61%); other responses included stable disease (13%), mixed responses/progression (27%) and missing (13%). PR/CR in individual organs (among those with organ involvement after excluding missing data) were as following: eye (12/32, 37%), mouth (9/17, 53%), esophagus (5/9, 55%), upper GI (2/3, 67%), lower GI (2/3, 67%), lung (1/9, 11%), and liver (5/14, 36%). 24% (95% CI 11.8-41.2%) had >50% reduction in glucocorticoid dose from baseline to 6 months. The cumulative incidences of treatment failure and NRM at 6 months were 15.2% (95% CI 6.6- 27.1%) and 2.2% (95% CI 0.2-10.3%). No patients have had relapse of malignancy. 6-month failure free survival was 84.8% (95% CI 70.7-92.4%) (Fig 2). Any grade and grade 3 or higher adverse events occurred in 55% (n=26) and 36% (n=17) of patients. 6 out of 17 grade 3 or higher adverse events were considered possibly or probably related to ruxolitinib. AE occurring in >10% of patients included dry eye, diarrhea, nausea, vomiting, infection, upper respiratory tract infection, weight gain, laboratory and other abnormalities. PRO analyses are ongoing and will be presented.
Discussion: The trial met its primary endpoint with half of adults with moderate-severe sclerotic cGVHD achieving partial responses in skin or joint as well as overall cGVHD responses. The use of ruxolitinib was associated with high rates of failure-free survival. In conclusion, the use of ruxolitinib results in high rate of responses in highly morbid sclerotic form of cGVHD.
Disclosures
Bhatt:National Marrow Donor Program: Research Funding; Pfizer: Research Funding; Servier Pharmaceuticals: Honoraria; Abbvie: Honoraria, Research Funding; Incyte: Honoraria, Research Funding; Genentech: Honoraria; Protagonist: Other: Member, Safety Monitoring Committee ; Jazz: Research Funding; Chimerix: Other: Drug support for a trial. Saad:In8Bio Inc: Patents & Royalties; CareDx: Consultancy; Incyte: Consultancy; Magenta Therapeutics: Consultancy; Kite/Gilead: Consultancy. Hamilton:Syndax: Membership on an entity's Board of Directors or advisory committees; Nkarta: Membership on an entity's Board of Directors or advisory committees; Incyte: Membership on an entity's Board of Directors or advisory committees; Equilium: Membership on an entity's Board of Directors or advisory committees; Kadmon: Membership on an entity's Board of Directors or advisory committees. Gundabolu:BioMarin Pharmaceuticals: Honoraria; BMS: Honoraria; Novartis: Honoraria; Blueprint Medicines: Honoraria; Samus: Research Funding; Jazz: Honoraria; Pfizer: Honoraria, Research Funding. Lee:Syndax: Research Funding; Equillium: Consultancy, Honoraria; Kadmon: Consultancy, Honoraria, Research Funding; Amgen: Research Funding; AstraZeneca: Research Funding; Incyte: Research Funding; Pfizer: Research Funding; Novartis: Other: Steering Committee; Amgen: Research Funding; AstraZeneca: Research Funding; Incyte: Research Funding; Kadmon: Consultancy, Honoraria, Research Funding; Pfizer: Research Funding; Syndax: Research Funding; National Marrow Donor Program: Membership on an entity's Board of Directors or advisory committees; Mallinckrodt: Consultancy, Honoraria.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal